This week I am at the Hitachi Central research Lab in Kokubunji, Japan, to attend Hitachi’s annual Kenpatsu. This is an event where the Central research lab provides an update to the global Hitachi organizations on the many projects that they are working on. Since Hitachi is a technology company with extensive R&D capabilities, you can imagine how exciting this was for me to attend especially with the explosion of technology that is going on today. The main focus for Hitachi’s research is on Social Innovation, making the world a smarter, healthier, and safer world.

One project that was especially interesting for me (considering my advanced age) was the work that was being done in regenerative medicine. This work is being done in our Kobe Lab, in research partnership with Kyoto University and Sumitomo Dainippon Pharma Co., Ltd. Kyoto University is where Dr. Shinyu Yamanaka was awarded the Nobel Prize for the discovery that mature cells could be converted to stem cells for use in regenerative medicine.
Regenerative medicine is a branch of research in tissue engineering and molecular biology which deals with the "process of replacing, engineering or regenerating human cells, tissues or organs to restore or establish normal function”.
The Hitachi Kobe lab is developing equipment to automate the production of Induced pluripotent stem cells or iPS cells which were first generated by Dr. Yamanaka. In 2006, Dr. Yamanaka established that by introducing a small number of genes into ordinary human somatic (differentiated) cells, these pluripotent cells can differentiate into any type of cell in the body and proliferate indefinitely in culture. The process of changing a cell from a differentiated to a pluripotent state is called reprogramming. The method developed by Dr. Yamanaka has been shown to be highly reproducible, relatively simple, and is considered a major scientific breakthrough. Currently, cell cultures are produced by hand, but only experts are capable of producing medical-grade quality cells. If medical-grade cells are only able to be cultured by certain skilled people, regenerative medicine will not become generally available. Hitachi would like to change that by developing automated culture equipment capable of the stable mass production of iPS cells.
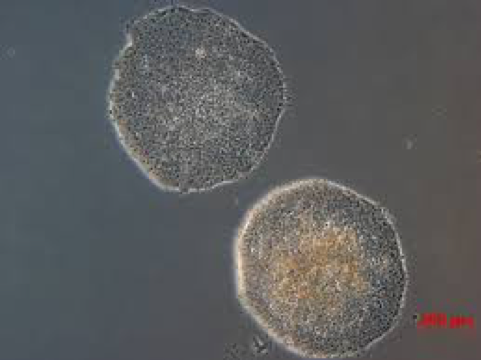
The prior alternative was to use human embryonic stem (ES) cells which were produced by removing cells from a 6-7 day old embryo and growing them in culture. While embryonic stem cells are natural, induced pluripotent stem cells can be generated using cells from an adult body, such as skin, which are plentiful and harmless to remove. As this does not require the destruction of an embryo, it avoids many of the ethical issues that surround human ES cells. Furthermore, unlike human ES cells, it is possible to derive patient-specific iPS cells and induce them into differentiated cells of various types, which can then be transplanted back into the patient without risk of immune rejection.
Hitachi began the research at their Center for Exploratory research in Saitama. Once they established the feasibility of building a machine to automate the development of iPS cells, Hitachi established the Hitachi Kobe Laboratory ("Kobe Lab") within the Kobe Biomedical Innovation Cluster ("KBIC"), where many people are carrying out cutting-edge advanced research in medical treatment, and moved the research team outside of the company in preparation for the social implementation phase. Hitachi felt that if they were to just stay within Hitachi, it would be difficult to develop a truly useful automated cell culture equipment. Hitachi believes in co-creation, that conducting R&D in the KBIC is the best environment to create truly useful automated cell culture equipment for the field of regenerative medicine. Hitachi Kobe Laboratory joined the Kobe Biomedical Innovation Center in the Kobe Biomedical Innovation Cluster.

Hitachi is developing automated culturing technology and process based on this research and clinical work that is capable of cultivating large quantities of high-quality medical grade cells for widespread use in regenerative medicine in the future. Hitachi has just overcome the first hurdle. This was to process cells with automated equipment that were of the same quality as those processed by expert human technicians. The next step is to be able to develop automated culture equipment capable of the stable mass production of cells of a quality exceeding that of the experts. The quality of the cultured cells is extremely important in regenerative medicine, and the "fight against bacteria" is a major issue. There are microorganisms in the air, and if just one of these bacteria enters the culture fluid, their presence will increase exponentially and destroy the human cell culture in an instant. In this event, those cells cannot be introduced to the body. This is a very difficult process and maintaining the sterility of the equipment is key when culturing cells.
Clinical research to confirm safety in humans commenced in 2013. For safety and other reasons, there is no fixed date, but researchers aim to make medical applications available as soon as possible. According to current research findings reported from Japan and overseas, iPS cells are capable of differentiation into the constituent cells of a wide range of tissues and organs, including nerves, cardiac muscle, and blood. (Think of the possibility of replacing brain cells that were damaged by Alzheimer) However, organs are more complex because of their three-dimensional (3D) structure. Small livers have been reported but there are as yet no reports of large 3D, functional organs of human size. This is an area that requires a combination of iPS cell technologies with 3D printers, biomaterials, and other technologies. This could be the next challenge for the Kobe Lab.
This was just one of many projects that I was able to hear about at our Hitachi Research Lab. If you are interested in hearing and seeing more of what Hitachi is researching for Social Innovation, you don't have to go to Kobe or Kokubunji. You can see them by visiting our NEXT 2019 event in Las Vegas, October 8-10 at the MGM Grand. You can click here to register.